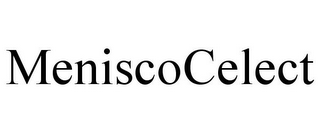CCI Trademark
Serial Number: 77213446 • Registration: 3589662
Trademark Classes
Class 1 - Chemicals
Chemical products for industry, science, photography, agriculture, horticulture and forestry
Class 5 - Pharmaceuticals
Pharmaceutical and veterinary preparations; sanitary preparations for medical purposes
Class 10 - Medical Apparatus
Surgical, medical, dental and veterinary apparatus and instruments
Class 42 - Computer & Scientific
Scientific and technological services; industrial analysis and research services
Class 44 - Medical Services
Medical services; veterinary services; hygienic and beauty care for human beings or animals
Owner Contact Info
Legal Representation
Trademark Details
Filing Date
June 22, 2007
Registration Date
March 17, 2009
Published for Opposition
December 30, 2008
Cancellation Date
October 23, 2015
Goods & Services
Pharmaceutical, veterinary or medical preparations for expansion, selection and implantation of human, in vivo, cartilage forming cells; and for controlling the quality of implants before they are implanted into patients; pharmaceutical and medical preparations, namely, nutritious media and substrates for expansion, selection and implantation of human, in vivo, cartilage forming cells including stem cell preparations or stem cell implants for controlling the quality of implants before they are implanted into patients; medical preparations, namely, polymer preparations and cell matrices of any type of origin for expansion, selection and implantation of human, in vivo, cartilage forming cells including stem cell preparations or stem cell implants for controlling the quality of implants before they are implanted into patients; test products for medical use, namely, nucleic acid probes, medical diagnostic reagents, antibodies for the purpose of the control of expansion, selection, and implantation of human, in vivo, cartilage forming cells including stem cell preparations or stem cell implants; hormones and means for the promoting of the growth of cells, namely, so called growth factors for medical use; enzymes for medical use; antibodies for medical use, for the purpose of expansion, selection and implantation of human, in vivo, cartilage forming cells including stem cell preparations or stem cell implants, and for controlling the quality of implants before they are implanted into patients
Preparations for scientific research, namely, nutritious media and substrates for expansion, selection and implantation of human, in vivo, cartilage forming cells including stem cell preparations or stem cell implants for controlling the quality of implants before they are implanted into patients
Pharmaceutical advice; veterinary and medical services; surgical implantation of human, in vivo, cartilage forming cells including stem cell preparations or stem cell implants; medical services featuring surgical follow-up
Veterinary or medical devices and instruments, also under the form of kits, for expansion, selection and implantation of human, in vivo, cartilage forming cells including stem cell preparations or stem cell implants for controlling the quality of implants before they are implanted into patients; medical devices and instruments, namely, biopsy kits, implantation kits, quality control kits for expansion, selection and implantation of human, in vivo, cartilage forming cells including stem cell preparations or stem cell implants for controlling the quality of implants before they are implanted into patients
Scientific research, development and study related to expansion, selection, quality control for others and implantation of human, in vivo, cartilage forming cells including stem cell preparations or stem cell implants; laboratory research related to the expansion, selection, quality control for others of human, in vivo, cartilage forming cells including stem cell preparations or stem cell implants; quality control for others of human, in vivo, cartilage forming cells including stem cell preparations or stem cell implants; assaying for the purpose of quality control for others of expanded or implanted human, in vivo, cartilage forming cells including stem cell preparations or stem cell implants; custom design and development of biochemical assays for the purpose of quality control expanded or implanted human, in vivo, cartilage forming cells including stem cell preparations or stem cell implants; scientific research, development and study related to the use of polymer membranes or cell matrices of any type of origin for expansion, selection, quality control for others of human, in vivo, cartilage forming cells including stem cell preparations or stem cell implant; pharmaceutical research services